
You are:
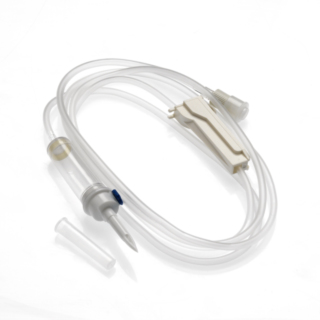
The INFINEED sterile, DEHP-free and BPA-free 1-way infusion device with photoprotector UV filter and Y-site has been designed to minimise its environmental impact, using only raw materials that are free from endocrine disruptors (elimination of components, including DEHP or BPA). The Y-site enables the bypass delivery of bolus drugs using a syringe and needle. Infuser with: Two-channel perforator Air intake: Delivered closed for immediate use on a 0.2µ filter bag. Chamber: Flexible and transparent, Calibrated at 20 drops = 1ml ± 0.1ml, 15µ particle filter. Flow regulator: Accurate and reliable, linear flow regulator. Tubing: PVC DINCH, transparent yellow, designed for comfort, optimal flow adjustment consistency and to prevent kinks. End fitting: Mobile Luer Lock End stopper: Equipped with a purge filter with anti-bacterial membrane.
Indications
This device enables the parenteral administration by gravity of injectable preparations: parenteral and photosensitive drugs, solutions and nutrition.
For peripheral lines, the infusion line can remain in place for a maximum of 96 hours (i.e. the maximum time the peripheral catheter can be in place).
For central lines, the infusion line must be changed at least every 72 hours (or according to the institution's validated protocol).
Used materials
PVC tubing chamber
DEHP < 0.1% m/m
ABS flow regulator
Luer lock ABS
Polyethylene (PE) protective cap
ABS and synthetic polyisoprene injection site.
Labeling
on each unit are written the reference, the lot/serial number, the sterilization method and the expiry date if applicable
medical device
sterile
Transport conditions
Protected from light, humidity and heat
| Conditioning | Parts quantities | Dimensions in cm | Weight in kg | Ean 13 of conditioning |
| UNIT | 1 | 3661809003417 | ||
| BOX | 100 | 39 x 30 x 28 | 3.400 | 3661809103414 |
| CASE | 400 | 60 x 41 x 59 | 15.000 | 3661809203411 |